The Real Danger of the NuvaRing
 Photo: Population Council / Hallie Easley
Photo: Population Council / Hallie Easley
By Rachel Walden — January 17, 2014
If you read the recent Vanity Fair article about NuvaRing contraception, “Danger in the Ring,” you could be excused for coming away with the conclusion that NuvaRing is deadly and should be yanked off the market immediately.
But hold on.
NuvaRing, a hormonal birth control that is inserted into the vagina, contains estrogen and etonogestrel, a form of progestin that’s different from what’s in the most common low-dose combined birth control pills. As the Vanity Fair article describes, it is also the subject of ongoing lawsuits about dangerous blood clots.
Marie Brenner’s article focuses heavily those lawsuits alleging harms from the device. What it doesn’t do is put the risk of harms from NuvaRing in the context of other hormonal birth control options, or discuss real magnitude of risk of any of these options.
All hormonal birth control with estrogen carries some risk of blood clots. These are referred to as venous thromboembolism, or VTE, and can be fatal. The risks of the most common combined oral contraceptives are quite small.
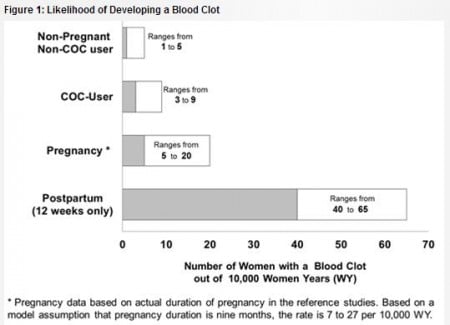
The latest FDA safety announcement (2012) estimates that for every 10,000 women taking birth control pills over the course of one year, between three and nine cases of blood clots may occur. In fact, the risks are thought to be higher during pregnancy (five to 20 cases per year for every 10,000 women), and much higher right after birth. By comparison, among non-pregnant women who do not take birth control pills, the number of cases per year ranges from one to five.
Some types of birth control, such as the newer types of pills (like Yasmin and Yaz) containing the progestin drospirenone, are thought to cause a greater risk. These pills have come under more scrutiny in Europe and the United States, with Europe taking a more active role in reviewing the risks. Some women’s health advocates, including Our Bodies Ourselves, have questioned allowing these newer pills to remain on the market because they pose a greater clot risk without offering any major benefits over older types of pills.
The factors to consider when weighing the risks and benefits of NuvaRing or other non-pill alternatives may be different, however, because non-pill alternatives offer more convenience and less chance of missing a dose.
Part of the problem in determining what’s “best” for any individual is the lack of clarity surrounding the increased risk that comes with using NuvaRing. Indeed, different studies have yielded different results: A BMJ study included in the Vanity Fair article found about a 90 percent increase in risk; other studies have reported “similar” rates of venous thromboembolism between the ring and the regular oral contraceptives.
Looking at the BMJ study, which seems most alarming, lets consider the actual numbers. The researchers explain a 90 percent increased risk would result in 7.8 incidents of VTE per 10,000 exposure years. That means, for example, if 1,000 women each used NuvaRing for 10 years, there would be about 8 incidents of VTE among them in that decade (1,000 women x 10 years = 10,000 exposure years).
So, while different methods of hormonal birth control carry different levels of risk, in general hormonal birth control is very safe for most women. Of great concern, however, is that women aren’t given this information to help in their decision-making.
A recent article in Huffington Post describes the FDA approval process for NuvaRing and alleges that Organon — the company that made NuvaRing, which pharmaceutical giant Merck now owns — maneuvered to keep clot risk information off the product label. To date, the NuvaRing label does not feature the same “black box warning” that the birth control patch carries, which notes a higher risk than the pill. Instead, the NuvaRing product label simply notes that smokers who use the ring may have “serious cardiovascular events.” The NuvaRing website does include some information on comparing the risk to combined oral contraceptives, but the FDA-approved label does not make this as clear by not including it in the black box warning that carries the most important safety information.
Cindy Pearson, executive director of the National Women’s Health Network, and a strong advocate for close review of drug safety, has responded to the concerns:
The most heartbreaking part of the Vanity Fair article are the accounts of women who never knew that the contraceptive ring was delivered a higher dose of hormones and is slightly more risky than pills. No clinician should offer women these products without fully disclosing the risks, and encouraging women to try alternative, safer forms of contraception if they haven’t already done so.
Agreed. While the risk to an individual woman may be low with any of these birth control methods, women must be informed that newer options — including the ring, the patch, and the newer pills — may be more risky than older-style combined oral contraceptives.
In addition, health care providers need to stay on top of the evidence, and should make a point of discussing the benefits and the risks. All women should have the information they need to make good choices for their own health.

Also don’t forget about MirenaIUD. This product has put many consumers in danger due to its SCARY side effects.
I know that contraceptives are very important for women and for the whole wide world! But why do these products kept on risking many lives?
Mark
http://mirenaiudlawsuitsinfo.com/
At the risk of sounding crass and harsh, I think the failure with this type, or any type, of birth control is the fault of the patient. I am lucky to have both a very good doctor and pharmacist, both of whom provided me with documentation and discussed potential side-effects and risks of the medication (NuvaRing) with me. However, it should be common sense that any time you place a chemical in your body (regardless of the potential benefits) there will ALWAYS be some form of risk. As the consumer/patient we are responsible to know what we are consuming and how it may harm us. I receive a booklet in each NuvaRing box that I get from the pharmacy that very clearly states there is an increased risk of blood clots with this medication. If you are literate, not knowing the risks of the medications you take is your own fault if you refuse to read the documentation that pharmacies are legally required to provide you with. Yes, I believe drug companies should do more to inform patients about the dangers of their medications. However the patient also has a responsibility to ask and research and take control of their own personal health care, or appoint a literate/intelligent person to help them.
If you are basing your “reading” the phamplet as a way to inform your self of the ricks associated with a drug, you are sadly mistaken. Use your literacy skills and read who is providing the FDA with the studies they use to publish the risks and side effects…….yes the drug companies themselves. Oh and better yet when the drugs are put up for review when people are hurt there is often a huge conflict of interest on the panel. For example, when 4 drugs made by Bayer (yaz and 3 others) were reviewed by an FDA panel, 3 on that panel have financial ties to Bayer and 1 who had voiced concern over the drug was excused from the panel. We are really making decisions based on the drug companies bottom line not on full-disclosure in the best interest of public safety.
Hiiiiii…. “literate friend”. You arrogant, snob of a person, you. For someone so presumptuous and full of self-confidence in relation to intelligence and competence, you should of all know-it-all’s be acutely aware of how idiotic it was to start your ignorant rant about consumers’ iignorance by saying “I hate to sound harsh or crass”, while knowing that was indeed exactly how you would sound. That, by the way, makes YOU sound awesomely ignorant yourself. And also…harsh and crass. You also began by stating that you are fortunate enough to have a wonderful doctor and pharmacist, knowing that so many others don’t and yet still insisting to go on with your bitch rant. Finally, you mentioned that lovely little pamphlet/insert included with your prescription. Not only does your ignorance know no bounds, because you were.clearly unaware of the multitudes of women living in small towns across America who are forced to fill prescriptions in terrible small pharmacies that often dont care enough to ensure that your medication include the proper warning labels and/or inserts(myself included-I had to look up my information online upon filling my 1st nuvaring prescription), but you must also be clueless to the fact that nuvaring does NOT concisely warn you on its pamphlet of the increased risk of blood clots. They found a sneaky way of dancing around the real warning, like so many other pharmaceutical companies do, and merely say some nonsnse that few women would interpret properly. So in the future, please refrain from being an uppity bitch.
To the “literate” commentor; I recently removed the Nuvaring after 4 day’s of use. I am a PhD in science, have conducted more research myself than most people have read, and am very familiar with what questions to ask and where to look. I too recognized that any hormonal contraceptive option posed some risk and deemed the potential benefit to outweighs the risk.
After 4 day’s with Nuvaring I suffered a psychotic break. I have 0 history of mental illness, depression, anxiety or suicidal thoughts and after just a few days of use I because physically and emotionally exhausted and began to question reality. Having no desire to end my life, I began wondering what would happen physically, if I engaged in certain dangerous acts. Not because I felt sad or wanted to hurt myself, because I became disassociated from reality. When I finally called a friend to Determine if she was a figment of my imagination or in fact a rthat al person I was taken to the ER. Luckily they asked about any new medications and when i informed the doc I was on the Nuvaring he told me to remove it immediately. Within just a few hours I began to feel normal again. The next day I could not believe what had happened to me the day before.
All this to say, I thoroughly researched this product before using it and nowhere in the literature that Nuvaring provides are these things mentioned. 99.9% of the risks written about are physical (blood clot, etc). I saw no warnings whatsoever about emotional or psychological affects and even asked my doc why they weren’t listed. She said given that the hormones are transmitted locally to the area where they are needed, it was likely that this product didn’t cause psychological side effects like an oral contraception can (which sounded like BS to me), but when the docs are saying this and you find no evidence otherwise, you make decisions differently than if you are completely informed. The only mention at all I saw was a note that said to inform a doc immediately if you have thoughts of suicide, which to be fair is written on every product ever made.
The ER doc told me this was not the first case he saw if this reaction, and it likely wouldn’t be the last. This product is made with third generation synthetic hormones which are less well understood than their predecessors and certainly transmission via the vagina is new and under researched. Simply put, there would have been no way of knowing about this risk at this point in time except via word of mouth from someone else who had experienced it themselves. So here you go ladies, I have and after only 4 day’s. I’m sure I’m In the very rare minority (as I tend to be with most reactions to meds), but none the less I could have easily hurt myself or someone else.
A few days out and I’m completely back to my normal thinking patterns, just exhausted and frustrated with the lack of info about this product.
[…] The Real Danger of the NuvaRing (Our Bodies, Ourselves) […]