Our Bodies Our Blog
- 2060
Celebrating 40 Years of the Black Women’s Health Imperative
October 30, 2023
On Friday, October 13, 2023, Black women and allies gathered at the Black-owned Salamander DC Hotel to celebrate the 40th Anniversary of…
Trusted Info on Perimenopause now available in Spanish and Armenian!
June 7, 2023
My own symptoms of perimenopause began while I was still getting a monthly period. My health care providers did not see them…

Abortion and Women’s Rights 1970
October 28, 2022
Two women tell the stories of their illegal abortions. More than 800,000 women every year sought out often unsafe abortions before the…

Our Bodies Ourselves Today Launch Celebration
October 25, 2022
On October 4, Our Bodies Ourselves Today held our launch celebration, hosted at Suffolk University. Please join us in the festivities by…

A message from the Founders of Our Bodies Ourselves
September 26, 2022
As we approach the 53rd anniversary of the first stapled, newsprint edition of Our Bodies, Ourselves, we are thrilled to announce and…

Welcome to Our Bodies Ourselves Today!
September 9, 2022
Dear Readers, We are thrilled to welcome you to Our Bodies Ourselves Today, a unique and trustworthy guide to your body and…

The Criminalization of Pregnancy
August 1, 2022
The following is an excerpt from Alice Rothchild’s op-ed, originally published in the Seattle Times. The recent Supreme Court decision to end…

Seize Back the Political Discourse on Life
July 6, 2022
For any woman who grew up as I did in the decades of expanding women’s rights, the majority opinion in Dobbs v…
Spreading the Word about Medication Abortion Via the Mail: An Urgent Priority for 2022
June 17, 2022
Our Bodies Ourselves is a long-time supporter of greater access to medication abortion via the mail and outside the formal medical system…

Invasions of Privacy: The Deluded Exception of Rape and Incest
June 15, 2022
by Alice Rothchild The following is an excerpt from Alice Rothchild’s op-ed, originally published in Common Dreams. One of the tools in…
May 20, 2022
by Alice Rothchild The following is an excerpt from Alice Rothchild’s op-ed, originally published in the Seattle Times. I came of age before…

The Legacy of Teen Voices Magazine
January 14, 2022
Teen Voices (1988-2012) was a trailblazing journalism, mentoring, and youth leadership nonprofit that produced an internationally distributed magazine written entirely by and…

Our Bodies Ourselves Update: November 2021
November 8, 2021
By the Our Bodies Ourselves Board of Directors: Diana Namumbejja Abwoye, Aziza Ahmed, Cris Alonso, Sherry Flashman, Miriam Hawley, Elizabeth Levy, Judy…
Watch Our Bodies Ourselves Virtual Events!
October 10, 2021
Each year, Our Bodies Ourselves participates in various events that feature or celebrate the history, legacy, and ongoing work of OBOS. This…

Celebrating “Nossos Corpos por Nós Mesmas” and Transnational Feminist Health!
October 1, 2021
After more than two years in the making, the first volume of “Nossos Corpos por Nós Mesmas,” has arrived! The book, a…

In the Fight Against HIV, Gendered Assumptions Have Deadly Consequences
August 19, 2021
In October 2019, the FDA approval of the new HIV-prevention drug Descovy was met with great excitement by doctors, HIV researchers, and…

Period Poverty is a Human Rights Issue. Period.
August 6, 2021
What is period poverty? Let’s talk about period poverty. Period poverty occurs when menstruators cannot access the menstrual products, underwear, and pain…

Stopping Keystone: A Victory for Native Women’s Health
July 1, 2021
In early June, after more than ten years of conflict, the Keystone XL oil pipeline project was halted by TC Energy, the…

Celebrate Pride Month with a #GiveOUTDay donation to Our Bodies Ourselves Today!
June 21, 2021
Our Bodies Ourselves Today, a new online platform featuring up-to-date information about health, sexuality, and well-being, is set to launch in 2022…

When Antidepressants Leave Lasting Damage: Living with Post-SSRI/SNRI Sexual Dysfunction
January 23, 2021
My clitoris is now no more than an inert and sensation-less nub of flesh. I am unable to feel attraction, arousal or…
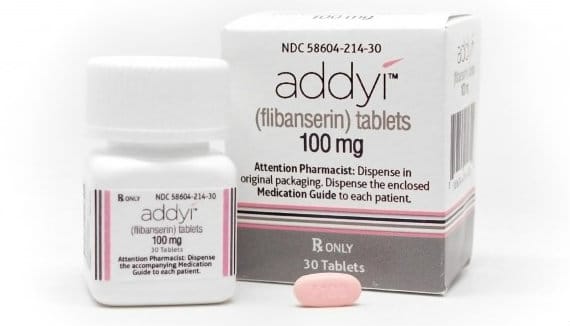
False or Misleading Claims: More Problems with Addyi
January 4, 2021
The so-called “pink Viagra” drug Addyi (flibanserin) is in the news again. This time, Sprout Pharmaceuticals, the drug’s manufacturer, is being called…